“Open the Flood Gates”
Since legalization of marijuana for recreational use in Missouri (11/8/22) and start of licensed sales (2/3/23), more Missourians have ready access to a wide variety of cannabis products. These legally-sold marijuana and marijuana-infused products are subject to “seed to sale” state regulations, including mandated 3rd-party lab analysis, that are intended to ensure cannabinoid content, quality, and safety in the name of public health.
However, there are other open sources of cannabinoid products that are not regulated, and therefore their content, quality, and safety are not assured. These are the internet sites and small-volume producers that supply the head shops/gas-station markets. They are in business by mis-application of the 2018 Federal “Farm Bill” that legalized hemp and its derivatives. In other words, other than Δ9-THC itself, if the cannabinoid is found in hemp they believe they can legally market it.
This has been the case with non-psychoactive CBD (cannabidiol), which is widely available and marketed as what can only be described as a “cure-all.” The FDA has only recently set its sight on CBD’s vast market, and will have a tough time reining it in.
Marijuana and Hemp, Research, and the Marketplace
Marijuana and hemp are essentially the same plant, Cannabis sativas/indica/ruderalis, but have lineages with different mixes of the main cannabinoids, Δ9-THC and the non-psychoactive CBD and CBC (Tetrahydrocannabinol, Cannabidiol, Cannabichromene). Over time, selective breeding has given rise to innumerable varieties and strains with different cannabinoid mixes, known as their profiles. Those high in THC are grown for human medical and recreational use, and those low in THC are grown for industrial fiber, CBD, and oil extraction. Targeted genetic engineering has already been used to hurry along the process of producing varieties with virtually no THC and high CBD content, and varieties with high THC content and not-much-else, to serve the different markets.
However, all Cannabis varieties have hundreds of identifiable cannabinoids, terpenes, and other chemicals, although most are present in only trace amounts. Research to isolate and identify these minor compounds is picking up in order to find individual components that may have specific medical applicability. In vitro and animal test systems can be used to identify, at the least, the affinity for the two human cannabinoid receptors and the isolate’s theoretical potency compared to Δ9-THC.
Organic synthesis of these compounds is also advancing. In fact, in several cases it was first synthesizing a specific compound in the lab that enabled the identification of the same compound in the plant. Once a specific cannabinoid can be synthesized, extracting its minute quantities from the plant is no longer necessary. The compound is still considered “a derivative” of Cannabis sativa, although it is not sourced from plants. Now available in quantity, it goes into the nebulous marketplace courtesy of the 2018 Farm Bill, and your patients might present with adverse effects after using some cannabinoid you’ve never heard of.
This is why the last few years have seen cannabinoid products purporting to contain Δ8-THC, Δ10-THC, Δ11-THC, THCP, THCV, CBN, etc. They are all found in Cannabis, and someone is able to obtain or produce sufficient quantity to sell.
A Light Touch of Cannabinoid Chemistry
Mother Nature can produce very complicated chemistry using a few basic building blocks and some well-designed enzymes.
As an aside, the cannabis plant synthesizes the corresponding carboxylic acid of each cannabinoid. This organic acid comes off with time or heat to yield the familiar compounds. For example, THC-A is Δ9-THC with a carboxylic acid side chain. THC-A is found in raw cannabis, which can be eaten, infused into an herbal tea, or juiced. However, it is converted to straight Δ9-THC when smoked or processed by extraction.
Many of the natural cannabinoids are variations in one or more of the 5 main regions of their core structure.
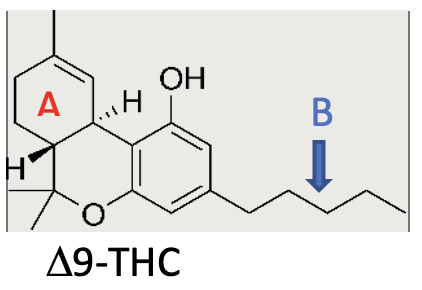
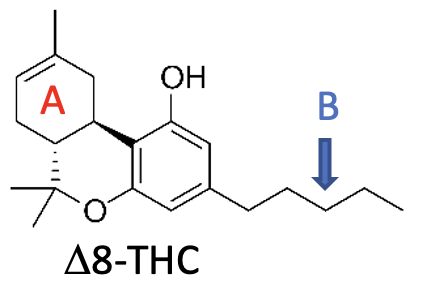
For example, as shown below, Δ8-THC is simply an isomer of Δ9-THC. The single double bond in the ring labeled “A” is in a different place. Δ8-THC has the same effects as Δ9-THC, but is 30-50% less potent.
Another example is the length of the side chain attached to the aromatic ring. In the structures above, both Δ9-THC and Δ8-THC have a 5-carbon side chain (labeled B). Similar cannabinoids have side chains varying in length from 3 to 7 carbons. The longer the side chain, the more potent the variant.
Variants with the 3-carbon chain are specified by the suffix “varin“, for example, TetraHydroCannabiVarin or THCV for short. Variants with a 7-carbon chain are specified by the suffix “phoral,” for example, TetraHydroCannabiPhoral or THCP for short.
Currently-Marketed Cannabis Derivatives
THCP (TetraHydroCannabiPhoral) is one to keep an eye on, because it is recognized as the strongest natural cannabinoid currently known. It has 30 times stronger affinity to the CB1 receptor and has a 5-10 times more potent psychoactive effect than Δ9-THC. As a comparison, a psychoactive dose of Δ9-THC is roughly 10-40 mg, whereas an equivalent dose of THCP is only 1-4 mg.
THC-0 (Tetrahydrocannabinol acetate ester) has an ester-linked acetate group on the fully-unsaturated ring of either Δ8 or Δ9-THC. In the structures above, the ring is not labeled but clearly has the only free-OH group. These acetate esters are said to be 3 times stronger than their corresponding THC and supposedly induce a more spiritual and psychedelic high. They also have a short delay in onset, thought to be due to necessary metabolism to some other active moiety.
HHC (Hexahydrocannabinol) is THC with no double bonds in the ring marked A above. It is fully hydrogenated with 6 hydrogens (“hexa”) instead of the 4 hydrogens (“tetra”) in Δ9-THC. Marketers claim it is 75% as potent at Δ9, but in vitro binding studies suggest much less activity.
CBN (Cannabinol) has mild sedative effects and is marketed as a sleep aide but has very little evidence to support it.
More are sure to come.
Any Relationship to the Original “Synthetic Cannabinoids” of a Decade Ago?
K2 agents, with brand names Spice, Incense, Krokadile and uncounted others were extremely potent and toxic agents that have largely decreased in popularity because of dramatic and unpredictable toxicity. They were first created to investigate the properties of the newly discovered cannabinoid receptors, but were resurrected for exploitation in the recreational drug market. Their structures bear no or little resemblance to the cannabis derivatives. The products’ chemical composition continually changed and became increasingly bizarre and effects unpredictable. No one really knew what they were buying. Effects included psychotic and affective disturbances, intense suicidal thoughts, self-harm behavior, and liver, kidney, and muscle injury. These adverse effects often took weeks to resolve and many did not resolve completely.
Why so toxic? It is likely because THC, the natural psychoactive cannabinoid found in recreational and medical marijuana, is only a partial agonist at CB1 receptors. Furthermore, other natural cannabinoids in marijuana, principally cannabidiol (CBD), act to modulate receptor activation by THC. In contrast, the K2 agents have full agonist activity at the cannabinoid receptors and are also orders of magnitude more potent than THC; and there is no modulating cannabinoid present.
These differences help explain the significant psycho-toxicity and other undesirable psychological and behavioral effects of the K2 agents that are largely absent in marijuana toxicity. The jury is out on the clinical outcome of the various natural cannabis derivatives, whether they will be more like marijuana or more like the K2 agents. Some of the cases in the literature are already highlighting unexpected toxicity.
A Cautionary Tale
If they’re derivatives of natural cannabis, what could go wrong? As said in the beginning of this Poison Alert, the Farm-Bill-enabled hemp sources of cannabinoid products are not regulated, and therefore their content, quality, and safety are not assured. The actual chemical composition and dose may be different from what is on the label.
As an example, the popular Δ8-THC is not extracted from the cannabis plant because there are only minor amounts. It is made by processing CBD (cannabidiol) which *is* produced in substantial quantities in many varieties of hemp. As with many organic syntheses, it is prone to side reactions, including production of Δ9 and Δ11-THC. A 2022 publication (Ray et al; Molecules) re-analyzed ten Δ8 commercial products and identified numerous impurities, even among those that had a Certificate of Analysis claiming nothing else was there. They identified three potential sources of problems – the use of impure CBD feedstock, poor post-reaction purification, and poor analytical practices during certification of purity. Production from low-budget marketers is likely to display even more variability in content, inaccurate quantitation, and potential toxicity.
This insight can be combined with clinical reports of toxicity from Δ8-THC products in children and teenagers. Despite the “less-potent” nature of this cannabinoid, encephalopathy and bradypnea requiring mechanical ventilation have occurred. Other less-severe effects, similar to Δ9-THC, include hypotension, tachycardia, dizziness, ataxia, sedation, and distorted vision. It is not yet known what role the Δ9-THC and other impurities are playing in Δ8-THC toxicity.
Clinically, the Cannabis Derivatives Are Handled the Same: Treat What You See.
In the end, it doesn’t really matter what version of the hemp-derived cannabis compounds have been used by the patient in front of you. There is no antidote; care is symptomatic and supportive for all of them. The urine drug screen might be positive for Δ9-THC, either due to actual Δ9-THC in the product or due to some degree of cross reactivity. Confirmation by GC-MS may or may not find the cannabis derivative(s), so history of the product used might be all there is to go on.
Mastering the ever-increasing spectrum of THC and cannabis derivatives is a daunting task. The Missouri Poison Center is here to help 24/7 by calling 1-800-222-1222. Our specially trained nurses, pharmacists, and toxicologist can offer information on THC and its various analogues, and provide the most up-to-date information regarding exposures and treatment.